CRO Business

Ark Medical Solutions Inc. (ArkMS), a subsidiary of Alfresa Holdings Corporation, provides support for development of pharmaceuticals, medical devices, and regenerative medicine products. Leveraging its more than 30 years of experience as a CRO*1, the company offers comprehensive services to pharmaceutical companies in various fields including clinical trials where pharmaceutical manufacturers, etc. administer new candidate drugs to humans to verify the safety and efficacy, trials led by physicians and researchers, and post-marketing surveillance that studies the side effects and efficacy after approval and launch of a drug.
- CRO (contract research organization): A specialized organization that supports the series of operations related to clinical trials carried out by pharmaceutical companies and research institutions.
Development of Ethical Pharmaceuticals
Ethical pharmaceutical development starts from basic research and targets the substance that causes the disease, identifies the effective drug ingredient, and develops the medicine. The safety and effectiveness of the drug are tested first in non-clinical trials on animals, before which clinical trials (phases I to III) are conducted on humans to study the efficacy and side effects in detail. The drug is then approved after review by the Pharmaceuticals and Medical Devices Agency (PMDA). Post-marketing surveillance is carried out after the product hits the market to continuously monitor the usage status.
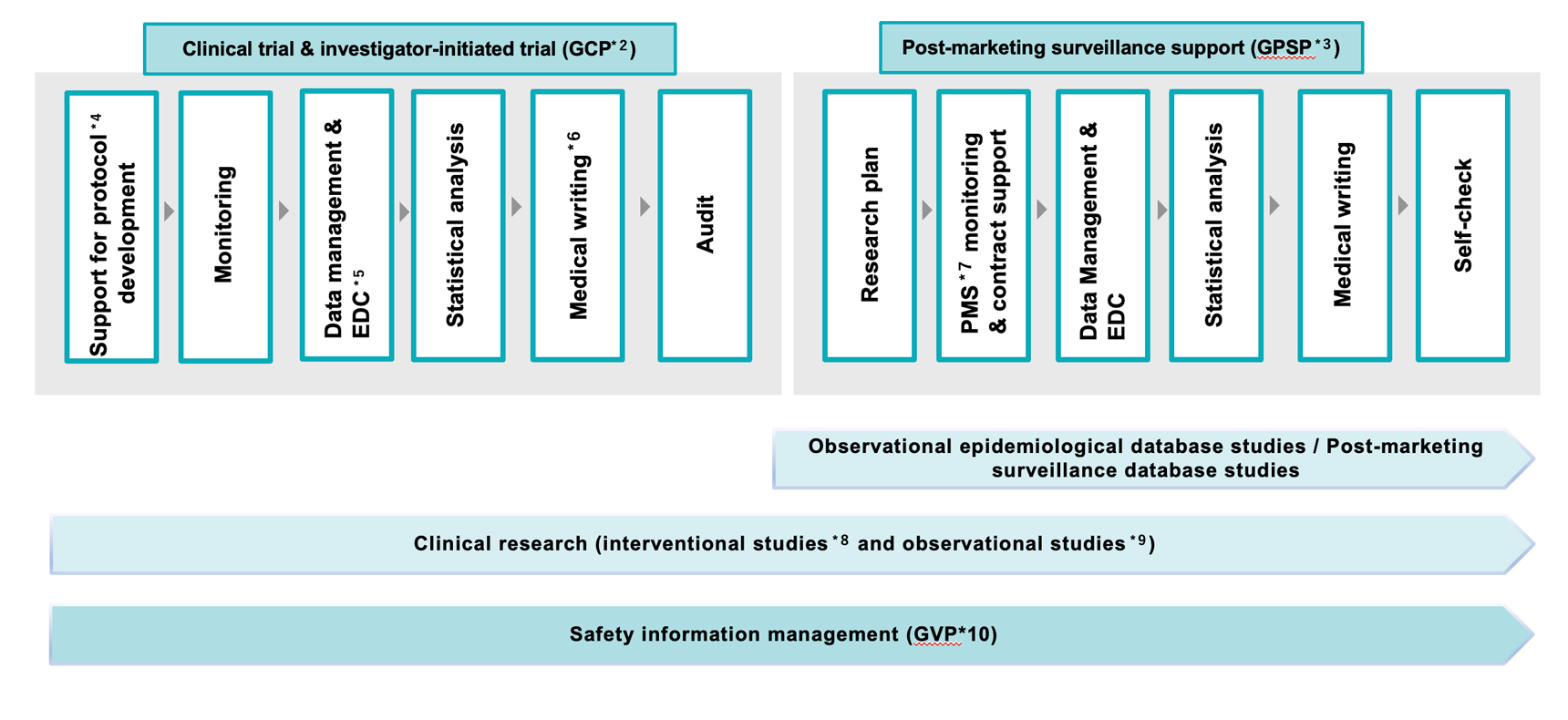
Service Area
- GCP: (Good Clinical Practice) A global standard and regulations for conducting clinical trials ethically and scientifically and ensuring the credibility of data
- GPSP: (Good Post-Marketing Study Practice) A Japan-specific ministerial ordinance that stipulates methods and procedures for use-results surveys, database studies, and other matters to verify the post-marketing safety of pharmaceuticals
- An official document detailing the plan for implementing a clinical trial, including objectives, design, evaluation items, and statistical analysis
- A system for collecting and managing case report forms electronically, rather than on paper
- The process of preparing specialized medical and pharmaceutical documents, such as clinical study reports, regulatory submission materials for drug approval, and academic conference presentation materials, while ensuring scientific validity and consistency
- Surveillance for ensuring the quality, efficacy, and safety of pharmaceuticals and medical devices after they are put on the market
- Studies in which researchers intervene, including with new treatments for study participants, regarding factors believed to be causally related to a disease, and verify the effectiveness of the intervention by comparing it with previous treatments and other factors.
- Studies in which, without intervening, researchers collect and analyze data on the health and disease status of study participants to obtain new insights
- GVP: (Good Vigilance Practice) An ordinance for collecting and examining safety management information on post-marketing pharmaceuticals and medical devices and implementing measures to ensure their safety